1,8-Dihydroxy anthraquinone (CAS NO : 117-10-2)
1,8-Dihydroxyanthraquinone, commonly known as Danthron or Chrysazin, is a naturally occurring dihydroxylated anthraquinone derivative that occupies a significant niche in both therapeutic medicine and industrial organic synthesis. Structurally related to the bioactive aglycones found in botanical laxatives like Senna and Rhubarb, it has been traditionally utilized as a stimulant laxative that increases the motility of the large intestine. Beyond its clinical history, it serves as a vital organic intermediate for the production of diverse vat dyes and pigments, where its specific phenolic arrangement allows for complex molecular coupling. In modern chemical research, it is valued as a scaffold for developing DNA-binding agents and fluorescent probes due to its planarity and unique electronic properties. By adhering to rigorous purity standards, 1,8-Dihydroxyanthraquinone enables the precise synthesis of high-value colorants and pharmaceutical research molecules.
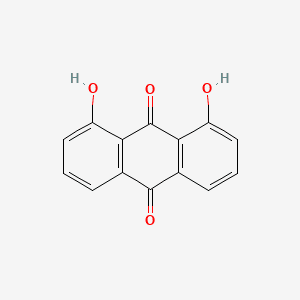
1,8-Dihydroxyanthraquinone, commonly known as Danthron or Chrysazin, is a naturally occurring dihydroxylated anthraquinone derivative that occupies a significant niche in both therapeutic medicine and industrial organic synthesis. Structurally related to the bioactive aglycones found in botanical laxatives like Senna and Rhubarb, it has been traditionally utilized as a stimulant laxative that increases the motility of the large intestine. Beyond its clinical history, it serves as a vital organic intermediate for the production of diverse vat dyes and pigments, where its specific phenolic arrangement allows for complex molecular coupling. In modern chemical research, it is valued as a scaffol...
Get a Quote
Details included in quote
Minimum Order Quantity
Lead Time
Regional Availability
Incoterms

Chemical Properties & Specifications
Identifiers of 1,8-Dihydroxy anthraquinone
Properties of 1,8-Dihydroxy anthraquinone
Safety & Hazards of 1,8-Dihydroxy anthraquinone
GHS Hazard Statements
H351 (96%): Suspected of causing cancer [Warning Carcinogenicity
Precautionary Statement Codes
P203, P280, P318, P405, and P501
Applications of 1,8-Dihydroxy anthraquinone
Stimulant Laxative
Clinical settings to treat constipation, particularly in palliative care, by stimulating the myenteric plexus to increase peristalsis
Oncology Research
KSM for the synthesis of anthracycline-like compounds and DNA-intercalating agents studied for their potential anti-tumor activity
Diagnostic ToolS
Development of specialized fluorescent chemosensors for detecting metal ions in biological systems
Vat Dye Intermediate
Serves as a critical precursor for the synthesis of alizarin-type dyes and other high-fastness organic pigments used in the textile and plastics industrieS
Analytical Reagent
Laboratory settings for the detection of aluminum and other beryllium-group metals due to its chelating properties
Surface Treatment
Specialized electrochemical processes to modify electrode surfaces for research in energy storage
Storage of 1,8-Dihydroxy anthraquinone
Store in a cool, dark, and dry area (15-25°C)
Keep away from strong oxidizing agents
Exposure to direct light can cause degradation of the crystalline structure and darkening of the color
Documents of 1,8-Dihydroxy anthraquinone
Have Questions About 1,8-Dihydroxy anthraquinone?
We've Got Answers.
In many jurisdictions (including the USA and parts of Europe), Danthron use is restricted to specific clinical cases, such as palliative care, due to concerns regarding its potential genotoxicity and long-term safety profile. It is largely replaced by Senna-based products for general constipation.
While both are dihydroxyanthraquinones, Chrysazin has hydroxyl groups at the 1 and 8 positions, whereas Alizarin has them at the 1 and 2 positions. This difference in substitution significantly changes their dyeing properties and clinical activity.
Small proprotions in various plants like Rhubarb and Aloe. However, for industrial and pharmaceutical use, it is typically synthesised for higher purity and consistency.
Phenolic hydroxyl groups deprotonate in basic conditions, resulting in a strong bathochromic shift (the color typically changes from orange to a deep violet or red), which is why it is often used as a pH-sensitive reagent.