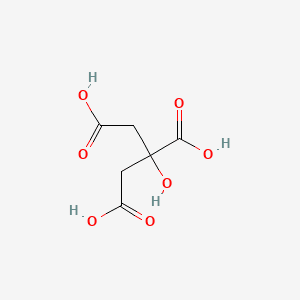
Citric Acid (CAS NO : 77-92-9)
Citric Acid (C₆H₈O₇) is a naturally occurring tricarboxylic acid found in citrus fruits, universally valued for its acidity, high solubility, and chelating properties. Citric Acid (2-Hydroxy-1,2,3-propane-tricarboxylic acid or E330) available in two main commercial forms: Citric Acid Monohydrate and Anhydrous Citric Acid, the difference being the presence or absence of water molecules in the crystalline structure. Both forms are supplied in IP/USP grades, ensuring high purity and consistent performance. Citric Acid serves multiple industries including Food & Beverage Manufacturing, Pharmaceuticals & Nutraceuticals, Cosmetics & Personal Care, Industrial Cleaning & Detergents, Chemical Manufacturing, and other specialized applications. We are among the trusted citric acid manufacturers and suppliers worldwide, offering high-quality monohydrate and anhydrous grades that meet quality and safety standards, with reliable lead times and global supply capability.
Citric Acid (C₆H₈O₇) is a naturally occurring tricarboxylic acid found in citrus fruits, universally valued for its acidity, high solubility, and chelating properties. Citric Acid (2-Hydroxy-1,2,3-propane-tricarboxylic acid or E330) available in two main commercial forms: Citric Acid Monohydrate and Anhydrous Citric Acid, the difference being the presence or absence of water molecules in the crystalline structure. Both forms are supplied in IP/USP grades, ensuring high purity and consistent performance. Citric Acid serves multiple industries including Food & Beverage Manufacturing, Pharmaceuticals & Nutraceuticals, Cosmetics & Personal Care, Industrial Cleaning & Detergents, Chemical Manufac...
Get a Quote
Details included in quote
Minimum Order Quantity
Lead Time
Regional Availability
Incoterms

Chemical Properties & Specifications
Identifiers of Citric Acid
Properties of Citric Acid
Safety & Hazards of Citric Acid
Applications of Citric Acid
Acidulant & Flavor Enhancer
Provides a sharp, tart taste in soft drinks, fruit juices, candies, and jams, balancing sweetness and enhancing overall flavor profiles
Natural Preservative
Acidic nature inhibits microbial growth, significantly extending the shelf life of various food products.
pH Adjuster & Buffer (Nutraceuticals, cosmetics, chemical manufacturing)
Crucial for maintaining stable pH levels in drug formulations, optimising stability, and efficacy. Essential for balancing the pH of creams, lotions, shampoos, and cleansers to match the skin's natural pH, ensuring product stability and skin compatibility. Used as a precise pH regulator in various chemical reactions and processes
Effervescent Agent
Reacts with bicarbonates to produce carbon dioxide, creating the fizz in effervescent tablets and powders, aiding in drug dissolution and palatability.
Chelating Agent
Bind strace metals that could otherwise catalyze degradation reactions in medicines
Antioxidant
Formulations from oxidation, maintaining product integrity.
Descaling
Effective Removal of mineral deposits (limescale, rust) from pipes, boilers, and industrial equipment due to its chelating properties.
Water Treatment
Controls pH and sequester metal ions in industrial water systems.
Plasticizers
Incorporated into some plastics to enhance flexibility