Pregabalin (CAS NO : 148553-50-8)
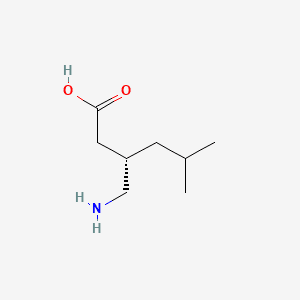
Pregabalin is the (S)-isomer of 3-isobutyl-gamma-aminobutyric acid. Therapeutic efficacy of Pregabalin is strictly dependent on its enantiomeric purity, as the (R)-enantiomer is biologically inactive. Pregabalin is an anticonvulsant therapeutic medication primarily used to treat neuropathic pain, fibromyalgia, seizures, and generalized anxiety disorder. 3-isobutyl GABA functions by binding to the alpha2-delta subunit of voltage-gated calcium channels in the central nervous system, inhibiting excitatory neurotransmitter release. As a blockbuster agent for Neuropathic Pain, Generalized Anxiety Disorder (GAD), and Partial Seizures, its manufacturing requires extreme stereochemical precision. We utilise world-class manufacturing infrastructure, employing either enzymatic kinetic resolution or asymmetric hydrogenative catalysis to achieve near-perfect stereoselectivity - free from chiral impurities - meets the most stringent global pharmacopoeial standards (USP/EP/BP/IP). Our high-performance Pregabalin API is a gold-standard gabapentinoid, meticulously synthesised for the global pharmaceutical sector as we are an established pregabalin api supplier with Pregabalin high purity. Our process chemistry is designed for low residual solvent profiles and a controlled impurity fate, making it the preferred choice for top-tier CDMOs and pharmaceutical formulators.
Documents
Pregabalin is the (S)-isomer of 3-isobutyl-gamma-aminobutyric acid. Therapeutic efficacy of Pregabalin is strictly dependent on its enantiomeric purity, as the (R)-enantiomer is biologically inactive. Pregabalin is an anticonvulsant therapeutic medication primarily used to treat neuropathic pain, fibromyalgia, seizures, and generalized anxiety disorder. 3-isobutyl GABA functions by binding to the alpha2-delta subunit of voltage-gated calcium channels in the central nervous system, inhibiting excitatory neurotransmitter release. As a blockbuster agent for Neuropathic Pain, Generalized Anxiety Disorder (GAD), and Partial Seizures, its manufacturing requires extreme stereochemical precision. We...
Get a Quote
Details included in quote
Minimum Order Quantity
Lead Time
Regional Availability
Incoterms

Chemical Properties & Specifications
Identifiers of Pregabalin
Properties of Pregabalin
Safety & Hazards of Pregabalin
Hazard Statements Code
H302, H315, H319
Precautionary Statements Code
P261, P264, P270, P280, P301+P312, P305+P351+P338 P304+P340: IF INHALED: Remove person to fresh air and keep comfortable for breathing.
Toxicological Summary
Limited information regarding overdose with pregabalin. The highest known accidental overdose of pregabalin during clinical development was 8000 mg; this event was without significant clinical consequences. The most commonly reported adverse events with overdose were reduced consciousness, confusional state, agitation, depression/anxiety, and restlessness in the postmarketing studies. In addition, heart block, seizures, and death have also been reported in some patients taking combination with other CNS depressants.
Overdose Management
Pregabalin can be removed by standard haemodialysis procedures, resulting in approximately 50% clearance of pregabalin in 4 hours.
Applications of Pregabalin
Neuropathic Pain Management
Pain treatment associated with diabetic peripheral neuropathy, postherpetic neuralgia, and spinal cord injury
Seizure Control
Adjunct therapy for partial-onset seizures and generalised anxiety disorder, enhancing overall seizure control.
Fibromyalgia Treatment
Provides relief from fibromyalgia-related pain, stiffness, and sleep disturbances.
Storage of Pregabalin
Storage Conditions
Store at 25 °C (77 °F); excursions permitted to 15 to 30 °C (59 to 86 °F). Maintain it in cool, dry, well-ventilated area, away from moisture and incompatible substances.
Packaging Options
Available in various sizes, including 25 kg double poly bags within HDPE drums for bulk applications.
Documents of Pregabalin
Have Questions About Pregabalin?
We've Got Answers.
Pregabalin is used to treat neuropathic pain, manage seizures, and alleviate symptoms of generalized anxiety disorder and fibromyalgia.
Pregabalin is typically administered as an oral capsule or solution. The dosage and frequency depend on the condition being treated and the patient's individual health needs.
Common side effects include dizziness, fatigue, weight gain, and swelling of the hands or feet. Patients should consult their healthcare provider if any side effects persist.
No, Pregabalin is a prescription medication, and it is available only through licensed healthcare providers.
(EXTRIP) suggests against performing extracorporeal treatment (ECTR)
Yes pregabalin is export ready with Pregabalin usp pharmacopeia grade attached with pregabalin coa, regulatory compliance documentation.