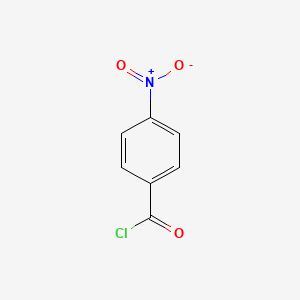
2,3,4,6-Tetrakis-O-trimethylsilyl-D-gluconolactone (Dapagliflozin Stage-3) (CAS NO : 32384-65-9)
The synthesis of CAS 32384-65-9 requires an inert atmosphere and anhydrous conditions to maintain the integrity of the trimethylsilyl (TMS) protecting groups. Our GMP manufacturing facility utilises advanced silylation protocols to ensure near-quantitative conversion and high chromatographic purity. This intermediate is engineered for high solubility in non-polar organic solvents, facilitating seamless coupling with organometallic reagents (such as Grignard or lithiated aryl species). By supplying a stabilised and vacuum-distilled product, we eliminate the variability often associated with in-situ silylation, thereby optimising the API scale-up yield and reducing downstream purification burdens. We are an established Dapagliflozin Stage-3 intermediate supplier, ensuring consistent high-purity Dapagliflozin intermediate. Being recognised as a CDMO partner for advanced pharmaceutical intermediates and a regulatory-compliant holder for global drug manufacturing chains, we provide a broad range of precursors with comprehensive analytical documentation (HPLC, GC-MS, NMR) and supporting quality assurance (Dapagliflozin intermediates COA/SDS/TDS). Our facilities are optimised for commercial-scale production and custom synthesis, offering a stable foundation for SGLT2 inhibitor manufacturing. We ensure reliable lead times and transparent project management, enabling partners to lead in high-precision pharmaceutical development.
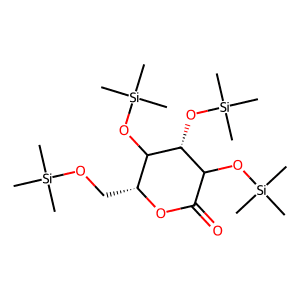
The synthesis of CAS 32384-65-9 requires an inert atmosphere and anhydrous conditions to maintain the integrity of the trimethylsilyl (TMS) protecting groups. Our GMP manufacturing facility utilises advanced silylation protocols to ensure near-quantitative conversion and high chromatographic purity. This intermediate is engineered for high solubility in non-polar organic solvents, facilitating seamless coupling with organometallic reagents (such as Grignard or lithiated aryl species). By supplying a stabilised and vacuum-distilled product, we eliminate the variability often associated with in-situ silylation, thereby optimising the API scale-up yield and reducing downstream purification burd...
Get a Quote
Details included in quote
Minimum Order Quantity
Lead Time
Regional Availability
Incoterms

Chemical Properties & Specifications
Identifiers of 2,3,4,6-Tetrakis-O-trimethylsilyl-D-gluconolactone (Dapagliflozin Stage-3)
Properties of 2,3,4,6-Tetrakis-O-trimethylsilyl-D-gluconolactone (Dapagliflozin Stage-3)
Safety & Hazards of 2,3,4,6-Tetrakis-O-trimethylsilyl-D-gluconolactone (Dapagliflozin Stage-3)
Applications of 2,3,4,6-Tetrakis-O-trimethylsilyl-D-gluconolactone (Dapagliflozin Stage-3)
Carbohydrate Protecting Group Chemistry
Highly stable, silyl-protected precursor that allows for regiospecific reactions at the anomeric centre without interfering with the secondary hydroxyl groups
GMP Analytical Standard
Serves as a high-purity volatile derivative for GC-MS sugar profiling, ensuring precise pharmaceutical QC/QA in complex carbohydrate characterisation.
C-Glycoside Precursor
Building block for the synthesis of Dapagliflozin, ensuring a streamlined pathway from D-Gluconolactone to the final bioactive molecule
Analytical Chemistry Standards
High-purity reference material for the validation of chromatographic methods in carbohydrate research
Regiospecific C-Glycosylation
Functions as a key electrophilic building block, where silyl protecting groups prevent unwanted side reactions at secondary hydroxyl sites during nucleophilic attack
Enabling Protecting Group Chemistry
Provides a robust TMS-protected matrix that facilitates purification and handling in large-scale API manufacturing environments
Storage of 2,3,4,6-Tetrakis-O-trimethylsilyl-D-gluconolactone (Dapagliflozin Stage-3)
Documents of 2,3,4,6-Tetrakis-O-trimethylsilyl-D-gluconolactone (Dapagliflozin Stage-3)
Have Questions About 2,3,4,6-Tetrakis-O-trimethylsilyl-D-gluconolactone (Dapagliflozin Stage-3)?
We've Got Answers.
Our high-purity 2,3,4,6-Tetrakis-O-trimethylsilyl-D-gluconolactone (Dapagliflozin Stage-3) is a benchmark product in our carbohydrate synthesis portfolio. As a specialized CDMO partner, we provide this derivatized lactone to facilitate the complex C-glycosylation steps required for SGLT2 inhibitor production.
In the SGLT2 synthesis pathway, any trace moisture will cause the trimethylsilyl (TMS) groups to deprotect prematurely. This leads to polymerization and significant yield loss during the C-glycosylation step. By delivering a vacuum-distilled product with ultra-low KF moisture, we provide the Technical Authority required for high-efficiency API scale-up, reducing the "Total Cost of Quality" for the innovator.
Many CDMOs struggle with de-bromination during the conversion of the ketone to the methylene bridge. Our team uses selective reduction chemistry that specifically targets the carbonyl group while leaving the C-Br and C-Cl bonds untouched. This level of chemical market intelligence allows us to provide a precursor that preserves the vital halogen functionalities required for the final coupling steps.
For commercial volumes, we offer a 10–12 week lead time, including full analytical validation (NMR, HPLC, GC-MS). As an experienced CDMO, we maintain a dedicated pilot plant to support the rapid transition from R&D quantities to metric-ton supply, ensuring your clinical and commercial timelines are never compromised.