

Trilaciclib Intermediate 2 (5-(4-(Propan-2-yl)piperazin-1-yl)pyridin-2-amine) (CAS NO : 866620-36-2)
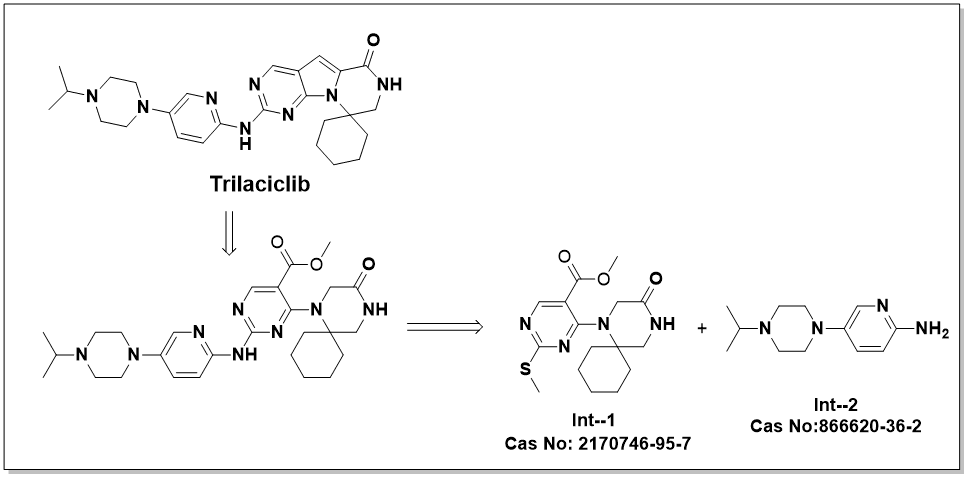
Our Trilaciclib API intermediate portfolio offers high-performance building blocks for the synthesis of the first-in-class, FDA-approved CDK4/6 inhibitor designed to protect bone marrow during chemotherapy. Trilaciclib (a kinase inhibitor used to reduce chemotherapy-induced myelosuppression) requires high-purity building blocks. Trilaciclib Advanced Intermediate 1 (CAS 2170746-95-7) & Trilaciclib Spiro-Core 2 (CAS 866620-36-2) represent critical milestones in the convergent synthesis of the spiro-pyrazino-pyrrolo-pyrimidine scaffold. Int -1 is synthesized through the condensation of 2-(methylthio)-7H-pyrrolo[2,3-d]pyrimidine-6-carboxylic acid and 1-aminomethyl-1-cyclohexanol. Int - 2 is then produced via a base-catalyzed internal cyclization of Intermediate 1, effectively locking the spiro-cyclohexane ring into the pyrazino-pyrrolo-pyrimidine framework. This "protection-free" methodology is preferred in 2026 for its high atom economy and reduced step-count compared to the original research routes. We utilise advanced spirocyclic construction and regioselective amination protocols to ensure these intermediates meet the stringent impurity profiles required for GMP-compliant drug substance manufacturing. Our manufacturing infrastructure is engineered for process safety and scalability, delivering high-purity solids with consistent (PSD). We are established global Trilaciclib intermediates supplier at 99%+ purity, ensuring minimal carry-over of methylthio-related impurities in the final API, and a broad range of various pharmaceutical intermediates for API manufacturers and semi-finished formulation suppliers, providing comprehensive analytical (HPLC/NMR/MS) and supporting quality assurance documentation (COA/TDS/SDS) across all grades, detailed impurity mapping; and region-specific regulatory support. We ensure reliable lead times from R&D quantities through commercial-scale manufacturing, enabling partners to meet diverse global and regional requirements.


Documents
Our Trilaciclib API intermediate portfolio offers high-performance building blocks for the synthesis of the first-in-class, FDA-approved CDK4/6 inhibitor designed to protect bone marrow during chemotherapy. Trilaciclib (a kinase inhibitor used to reduce chemotherapy-induced myelosuppression) requires high-purity building blocks. Trilaciclib Advanced Intermediate 1 (CAS 2170746-95-7) & Trilaciclib Spiro-Core 2 (CAS 866620-36-2) represent critical milestones in the convergent synthesis of the spiro-pyrazino-pyrrolo-pyrimidine scaffold. Int -1 is synthesized through the condensation of 2-(methylthio)-7H-pyrrolo[2,3-d]pyrimidine-6-carboxylic acid and 1-aminomethyl-1-cyclohexanol. Int - 2 is then...
Get a Quote
Details included in quote
Minimum Order Quantity
Lead Time
Regional Availability
Incoterms

Chemical Properties & Specifications
Identifiers of Trilaciclib Intermediate 2 (5-(4-(Propan-2-yl)piperazin-1-yl)pyridin-2-amine)
Properties of Trilaciclib Intermediate 2 (5-(4-(Propan-2-yl)piperazin-1-yl)pyridin-2-amine)
Safety & Hazards of Trilaciclib Intermediate 2 (5-(4-(Propan-2-yl)piperazin-1-yl)pyridin-2-amine)
Hazard Classes and Categories
Skin Irrit. 2 (100%), Eye Irrit. 2 (100%), STOT SE 3 (100%)
Precautionary Statement Codes
P261, P264, P264+P265, P271, P280, P302+P352, P304+P340, P305+P351+P338, P319, P321, P332+P317, P337+P317, P362+P364, P403+P233, P405, and P501
Applications of Trilaciclib Intermediate 2 (5-(4-(Propan-2-yl)piperazin-1-yl)pyridin-2-amine)
CDK4/6 Inhibitor Synthesis
CDK4/6 Inhibitor Synthesis: These intermediates are fundamental to constructing the complex spiro-structure of Trilaciclib, ensuring the correct spatial orientation for kinase binding. Oncology Drug Discovery: Essential for medicinal chemists developing next-generation myeloprotection agents and small-molecule inhibitors. Trilaciclib 1 & 2 intermediates are fundamental to constructing the complex spiro-structure of Trilaciclib, ensuring the correct spatial orientation for kinase binding Supply Chain De-risking: Our backward-integrated manufacturing ensures a steady supply of Int-1, reducing the complexity of multi-step API synthesis for pharmaceutical manufacturers.
Oncology Drug Discovery
Essential for medicinal chemists developing next-generation myeloprotection agents and small-molecule inhibitors
Process Chemistry R&D
Int-2 serves as the reactive chlorinated scaffold ready for palladium-catalysed cross-coupling or nucleophilic aromatic substitution (SNAr)
Supply Chain De-risking
Our backward-integrated manufacturing ensures a steady supply of Int-1, reducing the complexity of multi-step API synthesis for pharmaceutical manufacturers