Triphenylphosphine (CAS NO : 603-35-0)
Triphenylphosphine (PPh₃), also known as triphenylphosphane, is an organophosphorus compound featuring a central phosphorus atom bonded to three phenyl groups. Reagent and ligand in organic & organometallic chemistry: fundamentally, it functions as a nucleophile and a reducing agent due to the lone pair of electrons on the phosphorus atom, which is susceptible to oxidation. The compound’s versatility is unsurpassed in its role as a building block for Wittig reagents and as a key component in the Mitsunobu reaction and the Staudinger ligation. A ligand stabilises transition metal catalysts, governing their selectivity and reactivity, a critical function in large-scale industrial synthesis. Also referred to as Triphenylphosphane and Phosphine, Triphenyl Triphenylphosphine suppliers and manufacturers, respectively. We are the leading, trusted Triphenylphosphine supplier worldwide and in the United States, with reliable lead times and global supply capability.

Triphenylphosphine (PPh₃), also known as triphenylphosphane, is an organophosphorus compound featuring a central phosphorus atom bonded to three phenyl groups. Reagent and ligand in organic & organometallic chemistry: fundamentally, it functions as a nucleophile and a reducing agent due to the lone pair of electrons on the phosphorus atom, which is susceptible to oxidation. The compound’s versatility is unsurpassed in its role as a building block for Wittig reagents and as a key component in the Mitsunobu reaction and the Staudinger ligation. A ligand stabilises transition metal catalysts, governing their selectivity and reactivity, a critical function in large-scale industrial synthesis. Al...

.3d8f8f41.svg)
Industry
Elemental Derivatives
.3556d45a.svg)

Category
Phosphorus


Sub-category
Organo-Phosphorus Reagents & Ligands
Get a Quote
Details included in quote
Minimum Order Quantity
Lead Time
Regional Availability
Incoterms
.7767eb0f.png)

Chemical Properties & Specifications
Identifiers of Triphenylphosphine
Properties of Triphenylphosphine
Safety & Hazards of Triphenylphosphine
Safety Statements Code
S26-S36/37/39-S61
Risk Statements Code
R22; R43; R53
Hazard Declarations Code
H302; H317; H373
Caution Statements Code
P280; P301 + P312 + P330; P333 + P313
Applications of Triphenylphosphine
Pharmaceutical Intermediates & Precursors
Essential reagent in Mitsunobu reaction for forming complex esters and ethers, crucial steps in API synthesis (steroids, peptides)
Agrochemical Intermediates & Precursors
Synthesis of complex intermediates, certain organophosphorus pesticides & herbicides
Ligand
Preferred to stabilize and modify transition metal catalysts (e.g., Wilkinson's catalyst) for large-scale polymerization and fine chemical synthesis
Reagent
Wittig reaction for the stereoselective formation of carbon-carbon double bonds
Reducing Agent
To reduce azides to amines (Staudinger reaction) and hydroperoxides to alcohols
Polymerization Additive
Acts as a thermal stabilizer in certain polymer systems to prevent degradation during processing
Storage of Triphenylphosphine
Stability
Stable
Incomptability
Incompatible with oxidizing agents, acids
Condition
Store at RT
Documents of Triphenylphosphine
Have Questions About Triphenylphosphine?
We've Got Answers.
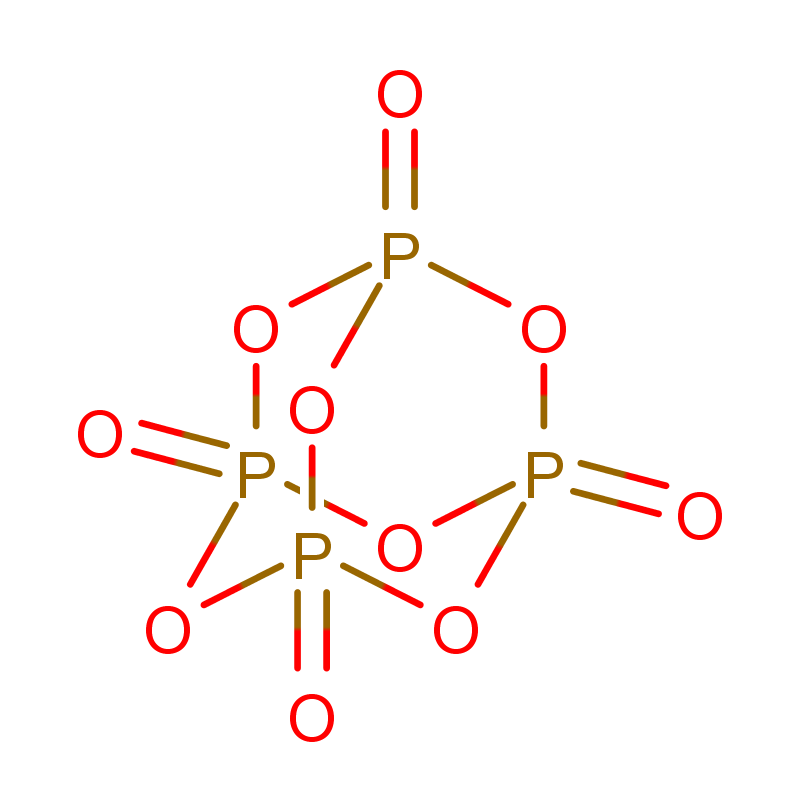
Byproduct is the often difficult-to-remove triphenylphosphine oxide. Strategies like phase separation or precipitation are employed in purification steps to remove this highly polar compound.
3 bulky phenyl groups make the phosphorus atom relatively large and polarizable, classifying it as a soft Lewis base, which favors bonding with soft transition metal ions (e.g., Rh,Pd,Pt) over hard ions.
Beacting PPh3 with an alkyl halide, forming a phosphonium salt, which is subsequently deprotonated by a strong base (e.g., n-BuLi) to yield the reactive ylide.
PPh3 oxidizes to OPPh3, which is inert in the Mitsunobu or Wittig reactions. Impurity levels as low as 1-2% can significantly impact yield and reaction consistency, necessitating a high-purity grade (99.5%) for complex API synthesis.
Typically not regulated as a controlled precursor, but its use in large quantities may require specialized disposal due to its organophosphorus nature.
Due to its excellent solubility, industrial reactions often utilize non-polar or moderately polar solvents such as Toluene, Tetrahydrofuran THF or Dichloromethane DCM.