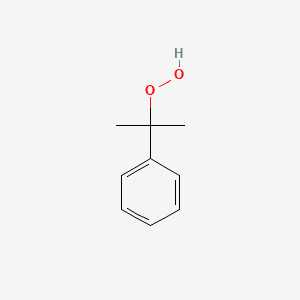
Cumene hydroperoxide (CAS NO : 80-15-9)
Cumene Hydroperoxide or Cumyl hydroperoxide is a clear to pale-yellow appearance characteristic, oily liquid classified as an organic peroxide, frequently used in chemical synthesis. Known as Percumyl H by supplier, primarily employed as an initiator for radical polymerisation and as an oxidising agent in industrial processes. With its ability to produce free radicals under mild thermal conditions, it plays deep critical role in the production of polymers, resins, and specialty intermediates. Cumene also acts as a Intermediate for plastics, and Oxidative Reactions for Commonly integrated into resin hardeners, composite materials, coatings, and epoxy formulations, and employed in oxidation-reduction setups for fine chemicals. Cumenyl hydroperoxide manufacturing has solvent compatibility, as it is soluble in ether, acetone, benzene, and aliphatic hydrocarbons.

Documents
Cumene Hydroperoxide or Cumyl hydroperoxide is a clear to pale-yellow appearance characteristic, oily liquid classified as an organic peroxide, frequently used in chemical synthesis. Known as Percumyl H by supplier, primarily employed as an initiator for radical polymerisation and as an oxidising agent in industrial processes. With its ability to produce free radicals under mild thermal conditions, it plays deep critical role in the production of polymers, resins, and specialty intermediates. Cumene also acts as a Intermediate for plastics, and Oxidative Reactions for Commonly integrated into resin hardeners, composite materials, coatings, and epoxy formulations, and employed in oxidation-re...

.3d8f8f41.svg)
Industry
Elemental Derivatives
.3556d45a.svg)

Category
Miscellaneous Functional Intermediates


Sub-category
General Functional Reagents
Get a Quote
Details included in quote
Minimum Order Quantity
Lead Time
Regional Availability
Incoterms
.7767eb0f.png)

Chemical Properties & Specifications
Identifiers of Cumene hydroperoxide
Properties of Cumene hydroperoxide
Safety & Hazards of Cumene hydroperoxide
GHS labelling
GHS02 (Flammable), GHS05 (Corrosive), GHS06 (Toxic), GHS03 (Oxidizing)
GHS Classification
Organic Peroxide (Type D) Acute Toxicity – Oral (Category 4) Skin Corrosion/Irritation (Category 1B) Eye Damage (Category 1) Specific Target Organ Toxicity – Single Exposure (Category 3, Respiratory Irritation)
Hazard Statements
*H226: Flammable liquid and vapor *H242: Heating may cause a fire *H302: Harmful if swallowed *H314: Causes severe skin burns and eye damage *H335: May cause respiratory irritation
Precautionary Advice
*P210: Keep away from heat/sparks/open flames *P280: Wear protective gloves/protective clothing/eye protection *P305+P351+P338: IF IN EYES: Rinse cautiously with water for several minutes
Environmental Notes:Environmental Notes: Environmental Notes
*Hazardous to aquatic life in concentrated form *Dispose as hazardous waste
Applications of Cumene hydroperoxide
Polymerization Initiator & Epoxidation Agent
Usage in the manufacturing of phenolic resins, styrene-butadiene rubber, and acrylics, as well as in oxidation of hydrocarbons like cumene to produce phenol and acetone.
High Radical Efficiency
Generates free radicals efficiently at ~60–120 °C
Low Freezing Point
Remains fluid at sub-zero temperatures (~–30 °C)
Controlled Decomposition
Predictable half-life for safe thermal use
Storage of Cumene hydroperoxide
Storage Temperature
2–8 °C, away from sunlight and incompatible substances
Shelf Life
6–12 months when kept in original, sealed containers
Packaging
UN-approved HDPE drums, 20–200 kg; temperature-monitored containers for transport
Handling Note
Use non-metallic tools; avoid contamination with reducing agents or heavy metals
Packing Group: II
Documents of Cumene hydroperoxide
Have Questions About Cumene hydroperoxide?
We've Got Answers.
It is insoluble in water but fully soluble in most organic solvents - ideal for non-aqueous polymerization or oxidation reactions.
Used at 0.5%–3% w/w depending on the reactivity of the monomer and the thermal schedule.
Its high reactivity with a moderate decomposition temperature allows faster cure cycles in epoxy resin and acrylic systems.
Always store under refrigeration, away from light and heat. Avoid contact with metal catalysts, strong acids, and reducing agents to prevent exothermic decomposition.
UN Classification: Class 5.2 (Organic Peroxide Type D, Liquid).
It has REACH & OSHA: Registered and regulated as a hazardous chemical; subject to risk assessments under process safety.