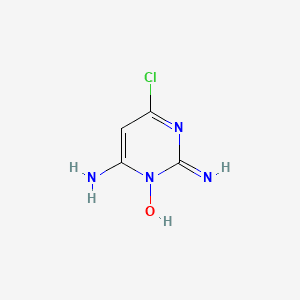
2,4-Diamino-6-Chloropyrimidine3-Oxide (CAS NO : 34960-71-9)
2,4-Diamino-6-Chloropyrimidine 3-Oxide is a pyrimidine derivative primarily used as a reference standard impurity for Minoxidil formulations. It plays a crucial role in impurity profiling and quality control within pharmaceutical manufacturing.

Documents
2,4-Diamino-6-Chloropyrimidine 3-Oxide is a pyrimidine derivative primarily used as a reference standard impurity for Minoxidil formulations. It plays a crucial role in impurity profiling and quality control within pharmaceutical manufacturing.

.3d8f8f41.svg)
Industry
Beauty & Personal Care
.3556d45a.svg)

Category
Hair Care Actives


Sub-category
Hair Regrowth & Scalp Treatment Actives
Get a Quote
Details included in quote
Minimum Order Quantity
Lead Time
Regional Availability
Incoterms
.7767eb0f.png)

Chemical Properties & Specifications
Identifiers of 2,4-Diamino-6-Chloropyrimidine3-Oxide
Properties of 2,4-Diamino-6-Chloropyrimidine3-Oxide
Safety & Hazards of 2,4-Diamino-6-Chloropyrimidine3-Oxide
Hazard Identification
GHS Classification: Not classified as hazardous under GHS.
(H-Statements)
H302: Harmful if swallowed (applicable under certain test conditions). H315: May cause mild skin irritation.
(P-Statements)
P264: Wash hands thoroughly after handling. P280: Wear protective gloves and eye protection. P301+P312: If swallowed, call a POISON CENTER or doctor/physician if you feel unwell.
Handling
Use PPE when handling the material. Avoid contact with skin and eyes.
First Aid Measures
Inhalation: Move to fresh air and seek medical attention if symptoms persist. Skin Contact: Wash with soap and water. Remove contaminated clothing. Eye Contact: Rinse cautiously with water for several minutes. Ingestion: Rinse mouth and seek medical advice.
Applications of 2,4-Diamino-6-Chloropyrimidine3-Oxide
Pharmaceuticals & Nutraceuticals
Used in the quality control and impurity profiling of Minoxidil APIs and formulations.
Beauty & Personal Care
Integrated into the quality assurance process for hair regrowth treatments and scalp health formulations related to Minoxidil-based products.
Storage of 2,4-Diamino-6-Chloropyrimidine3-Oxide
Temperature
Store at 2-8°C (refrigerated conditions).
Environment
Keep container tightly closed, dry, and away from incompatible materials.
Packaging Options
Available in 10 mg, 25 mg, and 100 mg packaging for laboratory and quality control use.
Documents of 2,4-Diamino-6-Chloropyrimidine3-Oxide
Have Questions About 2,4-Diamino-6-Chloropyrimidine3-Oxide?
We've Got Answers.
2,4-Diamino-6-Chloropyrimidine 3-Oxide is used as a reference standard impurity in the quality control of Minoxidil API and finished formulations. It helps ensure regulatory compliance by enabling accurate impurity profiling during pharmaceutical manufacturing.
This compound is commonly analyzed using high-performance liquid chromatography (HPLC), providing precise quantification in impurity profiling and stability testing of Minoxidil formulations.
The substance is generally stable under standard laboratory conditions but should be handled with care. Personal protective equipment (PPE) such as gloves, goggles, and lab coats are recommended, and handling should occur in a well-ventilated area to minimize exposure risks.
Monitoring this impurity ensures the safety, efficacy, and quality of Minoxidil-based pharmaceutical products. Controlling impurity levels helps prevent potential adverse effects and ensures compliance with global regulatory standards.